Using bacteria to target cancer treatment
When an outlet isn’t designed for your plug, you need a plug adapter to get power to your devices.
Researchers at the University of Cincinnati have developed a similar approach to cancer treatment, using bacteria as the adapter to connect powerful radiation therapy to cancer cells. The research was recently published in Advanced Healthcare Materials.
Radionuclide therapy basics
The research focuses on delivering a cancer treatment called radionuclide therapy. Nalinikanth Kotagiri, Ph.D., corresponding author of the research, explained that radioactive material used in the treatment releases beta rays as it decays, destroying anything in its path.
This treatment is typically delivered in a targeted way, with the cancer-killing radiation binding to the cancer cells through receptors on the cell surface to spare as many surrounding healthy cells as possible.
“Every cancer type has its own unique epitopes, or receptors, that are expressed on its surface,” said Kotagiri, a University of Cincinnati Cancer Center researcher and associate professor at UC’s James L. Winkle College of Pharmacy. “With that information, you can deliver a drug or radiation, using molecules such as antibodies and peptides, only to the cancer cells where it’s supposed to go, and not anywhere else like vital organs.”
Targeted therapy using conventional strategies can be effective, but cancer cells can mutate so that the receptors are no longer present on the surface and the radioactive material has nothing to bind to. This leads to treatment resistance, and the treatment can also be ineffective for other patients because they never had the receptors present on their cancer cells to begin with.
Following previous research that used probiotic bacteria to weaken cancer cell defenses, researchers in Kotagiri’s lab including lead study author Nabil A. Siddiqui asked if engineered bacteria could provide a target for radionuclide therapy when cancer cells have no receptors to target.
Bacterial adapters
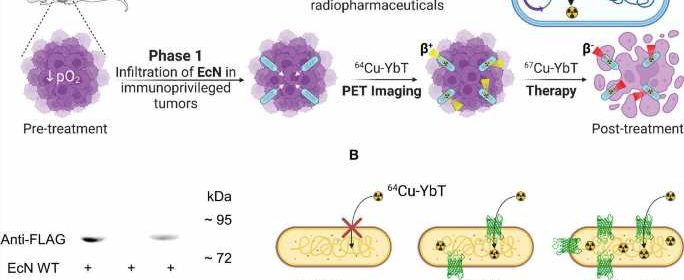
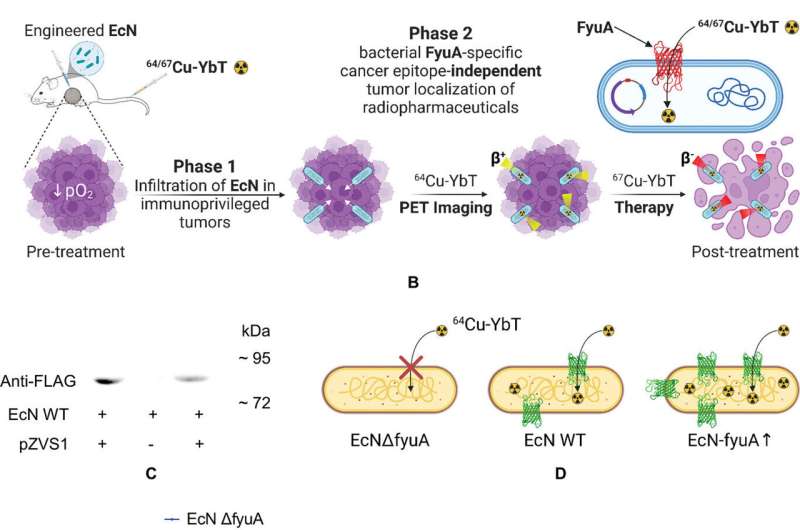
Kotagiri said probiotic bacteria can be helpful and are different from bacteria that cause diseases. While harmless to the cancer, bacteria tend to congregate inside tumors because they provide a low-oxygen, immunocompromised environment where bacteria thrive.
The research team engineered a unique strain of probiotic bacteria that over expresses a unique transporter that binds and concentrates metals, particularly copper, inside the bacteria. This process is facilitated by molecules known as siderophores that bind to metals.
Because the radionuclide is made of copper, the siderophore molecule latches onto it, making the targeted agent, which then binds to the bacterial transporter similarly to how antibodies bind to other receptors for traditional targeted therapy.
The engineered bacteria essentially provide artificial receptors for the targeted agent inside the tumors that can be effectively used as adapters in any tumor type, irrespective of the tumor’s receptor profile. The bacteria itself is highly resistant to radiation, but through a process known as the bystander effect, the radionuclide will destroy nearby cancer cells once it concentrates inside the bacteria.
“As long as these engineered bacteria are inside a tumor, these targeted agents, specific to the bacteria, will transport the radioactive metal,” Kotagiri said. “They won’t care if there is a cancer cell that is expressing a receptor or not. All they care is that they have identified something that they can recognize, accumulate and retain in.”
The ability of the engineered bacteria to sustain their population inside the tumors exclusively means that the bacteria do not have to be administered more than once.
Study results
Using animal models of colon and breast cancer, the research team injected the engineered bacteria directly into tumors. After waiting 48 hours for the bacteria to proliferate, they administered two doses of radionuclide therapy using copper-67, a high energy copper radionuclide.
The bacteria and radionuclide combination therapy led to nearly twofold survival compared to control models after 30 days. Kotagiri noted the concept was tested in a model of receptor negative breast cancer, and the therapy was effective in this treatment-resistant model.
Using a different low energy radioactive copper isotope, copper-64, Kotagiri said the same process can be used to image a tumor. The radionuclide still attaches to the bacteria populated inside the tumor, but instead of killing the cancer, it helps it “light up” on scans.
“So we can make this switch between copper-64 and copper-67 seamlessly to image the tumor, and then once we have imaged, we can introduce another molecule again to do therapy,” Kotagiri said. “We wanted to show that it’s possible to use the same kind of technology that was previously used for infection imaging for cancer therapy.”
Next steps
As research on bacteria-based therapies continues, Kotagiri said one hurdle will be gaining wider acceptance in the medical community that this approach is safe. More research will also need to confirm that the bacteria naturally and exclusively colonize inside tumors when delivered into the bloodstream.
Once a general consensus on safety is reached, Kotagiri envisions this therapy approach being tested in human clinical trials first in situations where the bacteria can be directly injected into tumors that are resistant, and where there are no other receptors or therapies available.
“Can this be something that can still make the targeted therapies work where patients with refractory or receptor negative cancer profile would receive this and not experience the side effects of conventional non-targeted therapies? Otherwise, the only other recourse is for the patient to receive these highly toxic drugs that can potentially cause significant morbidity,” Kotagiri said.
The research team is also testing the effectiveness of the approach using radionuclides already approved by the Food and Drug Administration in hopes that the technology can more quickly be tested on patients. Long term, Kotagiri hopes the process can be applied to tumors that are hard to image, access and treat.
More information:
Nabil A. Siddiqui et al, An Engineered Probiotic Platform for Cancer Epitope‐Independent Targeted Radionuclide Therapy of Solid Tumors, Advanced Healthcare Materials (2023). DOI: 10.1002/adhm.202202870
Journal information:
Advanced Healthcare Materials
Source: Read Full Article