Study raises possibility of immunotherapy treatment for ALS
New research reveals a type of monoclonal antibody already tested in certain forms of cancer may be a promising treatment in stopping the progression of amyotrophic lateral sclerosis, or ALS, a fatal neurodegenerative disease.
The study, led by scientists at Oregon Health & Science University, published today in the Proceedings of the National Academy of Sciences.
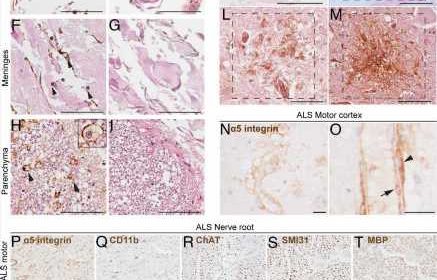
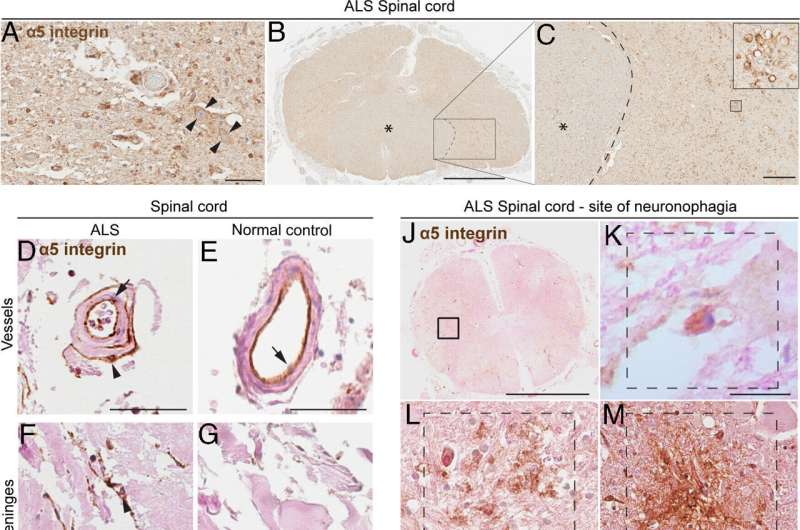
The study, involving a mouse model and confirmed in the tissue of human brains affected by ALS and donated after death, revealed for the first time that modulating immune cells can slow the progression of the disease. Previous research suggested a role for immune cells in ALS, but researchers this time used a high-throughput screening technique to identify a particular type of protein expressed on immune cells in the brain and spinal cord in people with ALS.
Researchers implicated the protein, known as alpha-5 integrin.
“When we blocked its expression in mice, we were able to slow down the disease,” said senior author Bahareh Ajami, Ph.D., assistant professor of molecular microbiology and immunology and behavioral neuroscience in the OHSU School of Medicine. “We hope that it will get to the clinic very soon.”
The team used a monoclonal antibody targeting a5 integrin, which had already been developed and used in treating certain forms of cancer. This means that it’s already undergone extensive safety studies to achieve approval through the Food and Drug Administration.
“Hopefully, it could be repurposed,” she said.
Using postmortem tissue from 139 brains donated for research, scientists confirmed the presence of a5 integrin within areas of the brain associated with motor function. Specifically, they found a5 integrin expressed by microglial cells and macrophages in blood—cells associated with the immune system—to be highly pronounced in the spinal cord, motor cortex and peripheral nerves during ALS.
They then tested the monoclonal antibody targeting a5 integrin in mice genetically predisposed to carry ALS and found that it protected motor function, delayed disease progression and increased mouse survival.
“We couldn’t believe they were doing so much better,” Ajami said.
Ajami, whose lab focuses on modulating the immune system to treat neurodegenerative diseases, said the study suggests the potential for applying immunotherapies to ALS as it’s already used in cancer and more recently through the use of monoclonal antibodies targeting Alzheimer’s disease.
“At this point, we cannot say it’s a cure but it’s a very interesting start,” she said. “It may be similar to what immunotherapy did for cancer or will do for Alzheimer’s by targeting immune cells.”
Ajami previously studied microglia in ALS. The study’s first author, Audie Chiot, Ph.D., of OHSU previously identified peripheral nerves macrophages as therapeutic targets in ALS mice. Today’s study complements their previous work by identifying a targetable protein on these cells.
Ajami came to OHSU in September of 2019, after beginning this line of research as a postdoctoral researcher at Stanford University. She said the next step in the research will be to develop dose response studies in the mouse model, and she ultimately hopes to see it progress to the point that it can be used to treat people with ALS.
In addition to Ajami and Chiot, co-authors on the study include Lawrence Steinman, M.D., Ph.D., of Stanford; co-first author Shanu F. Roemer, M.D., of the Mayo Clinic; Lisa Ryner and Michael Leviten of Pasithea Therapeutics; Alina Bogachuk, Katie Emberley, Dillon Brownell, Gisselle A. Jimenez, Randall Woltjer, M.D., Ph.D., of OHSU; and Dennis Dickson, M.D., of the Mayo Clinic.
More information:
Aude Chiot et al, Elevated α5 integrin expression on myeloid cells in motor areas in amyotrophic lateral sclerosis is a therapeutic target, Proceedings of the National Academy of Sciences (2023). DOI: 10.1073/pnas.2306731120
Journal information:
Proceedings of the National Academy of Sciences
Source: Read Full Article