Hard to drug: Protein droplets reveal new ways to inhibit transcription factors in an aggressive form of prostate cancer
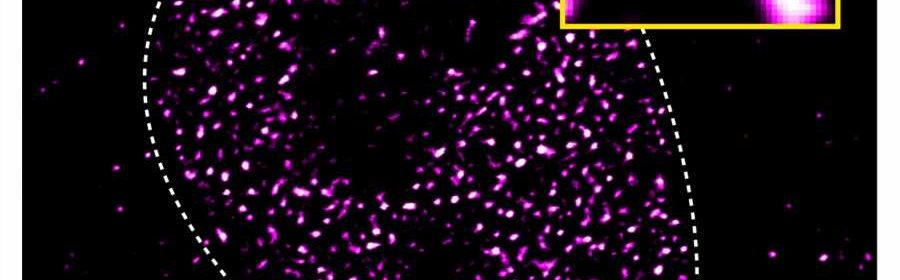
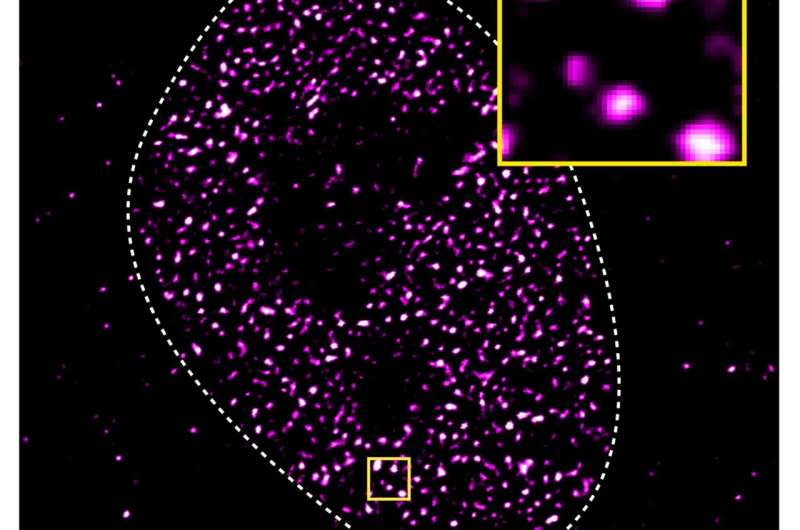
Transcription factors play essential roles in turning the genetic information encoded in genes into proteins in all cells and organisms. These regulatory proteins bind DNA, turn genes on or off, and control the rate at which DNA is transcribed into mRNA, which is needed for protein synthesis.
Because of their central role in transcriptional control, many diseases can be traced back to dysregulated transcription factors. Inhibiting their activity, especially in cancer, offers therapeutic potential, but many transcription factors have a trick up their sleeve. Their activation domains are intrinsically disordered, meaning that the chains of amino acids that make up the domain lack a clear three-dimensional structure. The lack of a stable 3D structure makes it virtually impossible to design drugs that bind to activation domains.
A research team led by Drs. Xavier Salvatella and Antoni Riera at IRB Barcelona, ICREA, and the University of Barcelona, Denes Hnisz at the Max Planck Institute for Molecular Genetics, and Marianne D. Sadar at BC Cancer (University of British Columbia, Canada)—focused on the tendency of intrinsically disordered proteins to form so-called biomolecular condensates. They found that the mechanisms involved in condensation could be exploited to inhibit androgen receptor activity in prostate cancer.
“The rationale we have followed to optimize an inhibitor of the androgen receptor could be exploited to inhibit other transcription factors, opening up new possibilities to address unmet medical needs,” says Dr. Salvatella.
Cellular droplets, a new approach to targeting transcription factors
Biomolecular condensates resemble proteinaceous blobs floating on water under a microscope. The condensates form in a process called liquid-liquid phase separation, similar to how oil droplets coalesce when mixed in water. “We had previously observed that the androgen receptor forms biomolecular condensates when you add even a tiny amount of an activating molecule, such as testosterone, to cells,” says Dr. Shaon Basu, now a computational biologist at the Charité and one of the study’s first authors together with Dr. Paula Martínez-Cristobal at IRB Barcelona.
The scientists hypothesized that there could be a link between the activation of the androgen receptor and its propensity to form droplets. Working with biophysicist Dr. Salvatella, they used nuclear magnetic resonance techniques to identify several short pieces within the intrinsically disordered activation domain that are essential for phase separation.
Moreover, the same short pieces were also necessary for the gene-activating function of the receptor. “We discovered short sequences in the activation domain that tend to be disordered when the protein is soluble, and surprisingly, these regions seem to form more stable helices when the protein is concentrated in condensates,” explains Dr. Hnisz. The short helices create transient binding pockets that can be targeted with inhibitors when the receptor is in condensates.
Improving compounds for the treatment of prostate cancer
Working with the labs of Dr. Antoni Riera and Dr. Marianne Sadar, the team then improved an experimental small molecule inhibitor to fit almost perfectly into the transient binding pocket. They then tested in cell and mouse models whether these changes would increase the efficacy in an aggressive, late-stage form of prostate cancer.
“We modified the chemical features of the compound to match the features of androgen receptor condensation, resulting in a tenfold increase in the potency of the molecule in castration-resistant prostate cancer,” says Paula Martínez-Cristobal, also the study’s first author. “This is really important because castration-resistant prostate cancer is an extremely aggressive cancer that is resistant to the current first-line therapeutics,” she adds.
However, more research is needed before these findings can be translated into new and safe therapeutics, the authors agree. The team hopes that the basic mechanisms they have discovered may be applicable to other transcription factors, opening the door to targeting these important molecules in many different diseases.
“We believe that the idea that there are certain sequences within intrinsically disordered protein domains that adopt a transiently stable structure in condensates is universal and likely generalizable to transcription factors,” concludes Dr. Hnisz.
The findings are published in Nature Structural & Molecular Biology.
More information:
Xavier Salvatella et al, Rational optimization of a transcription factor activation domain inhibitor, Nature Structural & Molecular Biology (2023). DOI: 10.1038/s41594-023-01159-5. www.nature.com/articles/s41594-023-01159-5
Journal information:
Nature Structural & Molecular Biology
Source: Read Full Article